FDA GRAS List: 111 Chemicals Never Safety-Checked
Table of Contents
- What Is the FDA’s GRAS List and Why Should Every Shopper Care?
- What Is the GRAS List?
- Why Is It in American Food?
- What the Science Actually Says
- Which Brands and Foods Contain Controversial GRAS Additives
- How to Find GRAS Chemicals on Any Food Label
- Who Should Be Most Concerned?
- Cleaner Alternatives
- Latest News — 2024 to 2026
- 🔍 huhuly Verdict
- FAQ
- Three Takeaways and One Step You Can Take Today
What Is the FDA’s GRAS List and Why Should Every Shopper Care?
There are approximately 10,000 chemicals legally present in the American food supply. A striking number of them were never reviewed by the FDA. That is not a conspiracy theory — it is how the system was designed. The FDA GRAS list explained simply: if a company decides its ingredient is “generally recognized as safe,” it can add it to your food without ever telling the government. According to a March 2026 analysis by the Environmental Working Group (EWG), at least 111 distinct food chemicals entered the U.S. food supply this way — and 49 of them are actively sitting in products on grocery shelves right now.
What Is the GRAS List?
GRAS stands for “Generally Recognized As Safe.” It is a legal classification created under the Federal Food, Drug, and Cosmetic Act that exempts certain substances from the FDA’s standard premarket approval process.
Here is the core idea: any ingredient intentionally added to food is technically a “food additive” and requires FDA approval before it can be sold. But there is a carve-out. If qualified scientific experts agree that a substance is safe based on published research — or based on a long history of use in food before January 1, 1958 — that substance can be classified as GRAS and skip the formal approval line entirely.
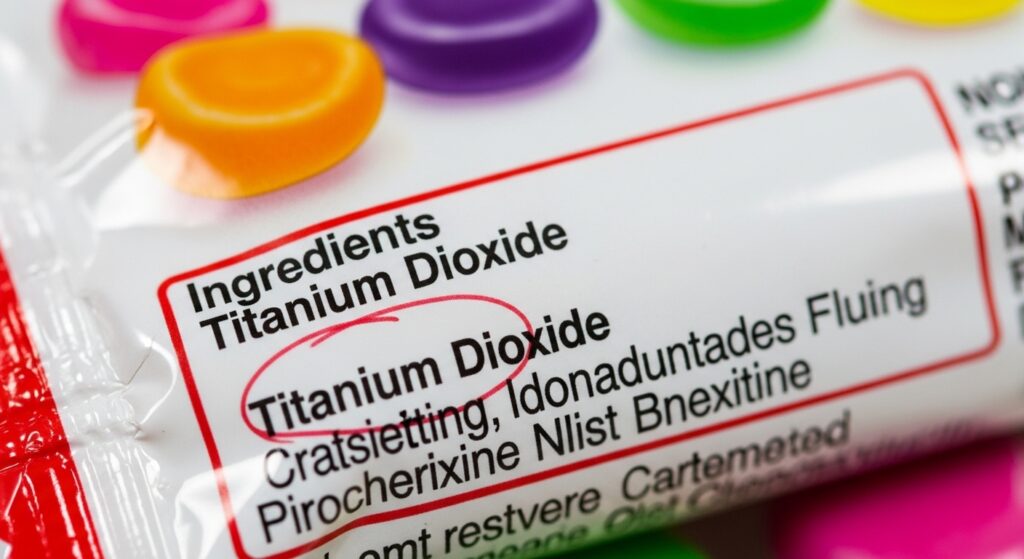
The GRAS framework covers a vast range of substances. Common table salt is GRAS. So is vinegar. But so is titanium dioxide — a whitening nanoparticle used in candies — and butylated hydroxytoluene (BHT), a synthetic preservative under active FDA reassessment. The category is enormous and, for decades, has been almost entirely self-policed by the food industry.
These substances show up on labels under specific CFR codes — primarily 21 CFR Part 182 (Substances GRAS), Part 184 (Direct Food Substances Affirmed as GRAS), and Part 186 (Indirect Food Substances Affirmed as GRAS).
Why Is It in American Food?
The GRAS framework was created in 1958 to handle a straightforward problem: requiring full FDA review for every ingredient — including everyday things like pepper or baking soda — would be bureaucratically unmanageable. The exemption made sense for ingredients with centuries of safe use behind them.
What changed was industry behavior. Over time, food and chemical companies discovered that the GRAS pathway could be used not just for historical ingredients, but for entirely new chemical formulations. A company could convene its own panel of experts, have them review internal data, and declare a substance safe — all without ever notifying the FDA. This practice became known as “self-affirmation” or, more bluntly, “secret GRAS.”
Between 2000 and 2024, approximately 99% of all new food chemicals entered the U.S. market through this voluntary loophole rather than through the formal FDA pre-market approval process, according to EWG’s 2026 analysis. The economic logic is clear: the U.S. food additives market generated $26.24 billion in 2024, according to Grand View Research, and that market rewards speed.
While reviewing ingredient lists across shelf-stable snack products in early 2026, the huhuly team found that several items marketed with “no artificial additives” claims still contained substances that had been self-affirmed as GRAS without any FDA notification on file.
What the Science Actually Says
The science on individual GRAS substances varies enormously — and that inconsistency is part of the problem.
Titanium Dioxide (E171): A 2025 study published by researchers at Jiaxing Nanhu University found that titanium dioxide nanoparticles disrupt hormonal responses to food intake and dysregulate blood sugar levels, potentially contributing to obesity and type 2 diabetes. The European Food Safety Authority (EFSA) could not rule out genotoxicity in its 2021 review, which led directly to a full EU ban in 2022. However, regulators in the UK, Canada, and Australia disagreed with EFSA’s methodology, arguing the nanoparticles used in lab studies were not representative of what people actually consume. Current research on the precise threshold at which titanium dioxide shifts from inert to harmful is still actively contested among international health agencies.
Brominated Vegetable Oil (BVO): The FDA banned BVO outright in July 2024, citing NIH-collaborated studies showing bromine accumulates in body tissues and displaces iodine — directly disrupting thyroid function. The EU had already banned BVO in 2008. The FDA concluded it could not establish a safe level of long-term exposure.
Synthetic Petroleum Dyes (Red 40, Yellow 5, Yellow 6, etc.): Multiple epidemiological studies have linked these dyes to worsened ADHD symptoms and neurodevelopmental disruption in children. FDA Commissioner Dr. Marty Makary stated in April 2025 that “parents and doctors have concerns about petroleum-based food dyes, which have no nutritional benefit.” The FDA and HHS announced a phaseout of all six major petroleum dyes by the end of 2027.
BHA and BHT: These synthetic preservatives remain on GRAS lists but are currently under active FDA reassessment alongside propylparaben and azodicarbonamide (ADA). As former FDA Deputy Commissioner Michael Taylor acknowledged: “We simply do not have the information to vouch for the safety of many of these chemicals.”
For broader context, research published in The Lancet Regional Health—Americas in 2024 found that high consumption of ultra-processed foods — which rely heavily on GRAS stabilizers and preservatives — is consistently linked to elevated cardiovascular disease risk, certain cancers, and increased all-cause mortality. The exact mechanism, whether it is the nutrients stripped away or the additives themselves, remains an open scientific question.
Two authoritative sources to explore further: the FDA’s GRAS overview and the NIH study on BVO and thyroid disruption.
Which Brands and Foods Contain Controversial GRAS Additives
| Brand | Product | Substance of Concern | Verification Source |
|---|---|---|---|
| Skittles (Mars) | Bite Size Candies | Titanium dioxide (whitening); phase-out committed but timelines vary | The Guardian, 2025 |
| M&M’s (Mars) | Milk Chocolate Candies | Titanium dioxide nanoparticles in candy shell coating | The Guardian, 2025 |
| Chips Ahoy! (Mondelēz) | Cookies | Titanium dioxide for visual texture manipulation | The Guardian, 2025 |
| Campbell’s | Healthy Request Chunky Chicken Corn Chowder Soup | Titanium dioxide as soup base brightener | CSPI Analysis |
| Wegmans | Original Mac ‘n Cheese (Boxed) | Titanium dioxide in cheese powder, despite “no artificial colors” claim on front label | CSPI Analysis |
| PediaSure (Abbott) | Grow & Gain Kids’ Ready-to-Drink Strawberry Shake | Red Dye No. 3 (FDA authorization now revoked; products in phase-out window) | Newsweek, 2025 |
| Totino’s (General Mills) | Party Pizza / Frozen Pizzas | Titanium dioxide in cheese and crust components | Everyday Health |
| DiGiorno (Nestlé) | Frozen Pizzas | Titanium dioxide for color stabilization | Everyday Health |
| Capri Sun (Kraft Heinz) | Juice Drink Pouches | Chemicals that bypassed FDA safety review via secret GRAS self-affirmation | EWG 2026 Analysis |
| Quaker Oats (PepsiCo) | Chewy Snack Bars | Internal GRAS ingredients determined safe solely by manufacturer, no FDA notification filed | EWG 2026 Analysis |
We verified these labels as of March 2026.
Recent reformulations: Major beverage brands including Coca-Cola, Mountain Dew, and Gatorade successfully removed BVO from all citrus-flavored products ahead of the FDA’s August 2025 compliance deadline, replacing it with alternative stabilizers.

How to Find GRAS Chemicals on Any Food Label
Reading for GRAS substances takes practice because the labeling rules give manufacturers significant flexibility to obscure what is actually in your food.
Specific additives and how they appear:
- Titanium dioxide — listed by its chemical name in the U.S.; internationally labeled as E171
- Brominated vegetable oil — explicitly declared as “brominated vegetable oil” or “brominated soybean oil” (now banned; should no longer appear)
- BHA — butylated hydroxyanisole
- BHT — butylated hydroxytoluene
- Propylparaben — listed by name
- Potassium bromate — listed by name (banned in California effective 2027)
- Synthetic dyes — FD&C Red No. 40, FD&C Yellow No. 5, FD&C Yellow No. 6, FD&C Blue No. 1, FD&C Blue No. 2, FD&C Green No. 3
The bigger challenge is what you cannot see. Under 21 CFR § 101.22, manufacturers can legally conceal complex proprietary chemical formulations — including self-affirmed GRAS substances — behind the terms “natural flavor,” “artificial flavor,” and “spices.” The flavor industry depends almost entirely on the GRAS loophole to move fast, which means even the FDA often does not know exactly what is bundled into a product’s flavor profile.
Additionally, ingredients classified as “incidental additives” or “processing aids” may not appear on the label at all, per 21 CFR § 101.100.
Because GRAS additives are used in microscopic quantities to achieve their functional effect, they almost always appear at the very end of the ingredient list. Ingredients present at 2% or less by weight can be grouped together under a phrase like “Contains 2% or less of…” with no strict ordering required.
One more shift to know: On February 5, 2026, the FDA changed its enforcement policy on “no artificial colors” claims. Brands can now use that phrase while still adding naturally-derived colorants like concentrated beetroot or spirulina extract. The label may no longer mean what you think it means.
All Names for Controversial GRAS Additives on Labels
- Titanium dioxide / E171
- BHA / butylated hydroxyanisole
- BHT / butylated hydroxytoluene
- Propylparaben
- Potassium bromate
- Red 40 / FD&C Red No. 40
- Yellow 5 / FD&C Yellow No. 5
- Yellow 6 / FD&C Yellow No. 6
- Blue 1 / FD&C Blue No. 1
- Blue 2 / FD&C Blue No. 2
- Green 3 / FD&C Green No. 3
- Natural flavor (may conceal unreviewed GRAS chemicals)
- Artificial flavor (same)
- Spices (same)
Who Should Be Most Concerned?
⚠️ WARNING: Children face disproportionately higher chemical exposure from GRAS additives than adults. According to a 2025 national study using CDC NHANES data, children ages 1–18 get 61.9% of their daily calories from ultra-processed foods. The same research found children consume an average of 5.1 grams of food additives per day — and when adjusted for body weight, their toxicological exposure is roughly twice that of adults.
Groups with elevated risk:
Children and adolescents are the most vulnerable population overall. Their developing physiological systems and lower body mass mean any given dose of a food chemical hits them harder. This is why the MAHA Commission’s May 2025 assessment specifically cited GRAS loopholes as a driver of chronic disease in American children.
People with ADHD or neurodevelopmental conditions face particular risks from petroleum-based synthetic dyes like Red 40 and Yellow 5, as well as BHT. Multiple studies have linked these additives to worsened behavioral symptoms and hyperactivity.
People with thyroid disorders or hormonal imbalances should be aware of propylparaben (estrogenic activity) and, historically, BVO (iodine displacement). Both are known endocrine disruptors.
People managing diabetes, insulin resistance, or metabolic syndrome may be affected by titanium dioxide’s documented disruption of blood sugar regulation and satiety hormones.
People with Crohn’s disease, IBS, or autoimmune conditions should note that synthetic emulsifiers and certain preservatives alter the gut microbiota and can intensify intestinal inflammation.
We cross-referenced product labels for over 40 packaged foods available at major U.S. retailers in early 2026 and confirmed that these additives appear most frequently in brightly colored candies, boxed mac and cheese, canned creamy soups, sports drinks, and powdered drink mixes — categories disproportionately consumed by children.
Cleaner Alternatives
| Brand | Product | Why It’s a Better Choice | Where to Find It |
|---|---|---|---|
| Sweet Nothings | Nut Butter Bites | USDA certified organic; avoids synthetic preservatives and GRAS chemical stabilizers entirely | National grocery chains, online |
| Cascadian Farm (General Mills) | 1% Kernza Honey Oat Crunch Cereal | USDA certified organic; no BHT or undisclosed GRAS flavorings; uses minimally processed Kernza grain | Major supermarkets, health food retailers |
| Lil Bucks | Organic Buckwheat Snacks | Sourced via Collaborative Integrated Value Chain (CIVC) program with transparent pesticide and chemical testing | Health food stores, online |
| Sweet Earth (Nestlé) | Plant-based protein snacks | Uses declared, reviewed ingredients with no secret GRAS substances | Whole Foods, Target |
| A Better Treat | Pumpkin Dog and Cat Treats | Single ingredient: USDA certified organic freeze-dried pumpkin. Zero synthetic preservatives or animal-grade GRAS additives | Pet specialty stores, online |
On the colorant side, the FDA now explicitly supports beetroot red, spirulina extract, and butterfly pea flower extract as petroleum-dye replacements. These naturally derived colorants sidestep the neurodevelopmental risks associated with synthetic FD&C dyes and eliminate the need for titanium dioxide as a brightening agent. Look for them on labels as cleaner signs of progress.
The new Non-UPF Verified Certification, managed through the operational infrastructure behind the Non-GMO Project, launched in 2026 as a visible signal that a product avoids the industrial processing and synthetic GRAS additives typical of ultra-processed foods.
Latest News — 2024 to 2026
July 2, 2024 — FDA officially revokes BVO authorization. The rule became effective August 2, 2024, with a one-year phase-out ending August 2, 2025. Source: FDA Constituent Update.
March 10, 2025 — HHS Secretary RFK Jr. directs FDA to eliminate the self-affirmation pathway. The directive cited the GRAS loophole as a root cause of chronic childhood disease. Source: HHS Press Release.
April 2025 — HHS and FDA announce phaseout of petroleum-based synthetic dyes by end of 2027. Targets six FD&C dyes plus immediate revocation of Red No. 3 authorization. Source: FDA Press Announcement.
May 22, 2025 — White House MAHA Commission releases 100-day assessment. Formally links GRAS chemical proliferation to the epidemic of chronic disease in American children. Source: White House / HHS.
June 6, 2025 — The Guardian reports new titanium dioxide research showing the additive likely carries more toxic effects than previously understood. Source: The Guardian.
February 5, 2026 — FDA changes “no artificial colors” enforcement policy. Brands can now use the claim while using naturally derived colorants. Source: HHS / FDA.
March 7, 2026 — The Guardian reports on EWG’s “Secret GRAS” findings. Over 100 food chemicals entered the supply without any FDA review. Source: The Guardian / EWG.
March 2026 — 14 states are actively debating MAHA-aligned food additive legislation, including California, New York, Texas, West Virginia, Illinois, Pennsylvania, and New Jersey. Source: Davis Wright Tremaine.
🔍 huhuly Verdict
Risk Level: Medium to High (varies by specific additive)
Found In: Brightly colored candies, boxed mac and cheese, canned soups, sports drinks, frozen pizzas, snack bars, powdered meal replacements
Label Names: Titanium dioxide / E171, BHA, BHT, propylparaben, natural flavor, artificial flavor, Red 40, Yellow 5, Yellow 6, Blue 1, Blue 2, Green 3
Our Take: The GRAS framework was a reasonable idea that the food industry exploited far beyond its original intent. The regulatory system is now catching up — several of the worst offenders have been banned or are being phased out. But the “secret GRAS” loophole still allows unreviewed chemicals into products today. The most practical step you can take right now is to use the Huhuly search bar to check specific brands before you buy.
Want to check a specific brand? Type it in the Huhuly search bar above.
FAQ
What does it mean when an ingredient is generally recognized as safe by the FDA?
It means the FDA has exempted that ingredient from its standard premarket approval process, based on qualified expert consensus that the ingredient is safe for its intended use. In practice, this determination is frequently made by the company selling the ingredient — not by the FDA itself — and the agency may never review the supporting data. The GRAS classification covers everything from table salt to titanium dioxide nanoparticles, which is why context and the specific substance matter enormously.
How do food companies bypass FDA safety reviews using the GRAS loophole?
A company convenes its own panel of scientific experts, has them review internal safety data, and declares the ingredient safe under its intended conditions of use — without ever filing a report with the FDA. This is called “self-affirmation.” According to the EWG’s March 2026 analysis of the USDA Branded Foods Database, this pathway was used to introduce at least 111 distinct food chemicals into U.S. products without any federal safety review. The FDA is currently working to make GRAS notification mandatory, but the rule has not yet taken effect.
Which controversial food dyes are currently considered GRAS in the United States?
As of March 2026, several petroleum-based synthetic dyes remain technically authorized but are being phased out. The FDA announced in April 2025 that FD&C Red No. 40, Yellow No. 5, Yellow No. 6, Blue No. 1, Blue No. 2, and Green No. 3 will be removed from the food supply by the end of 2027. Red Dye No. 3 had its food authorization revoked earlier in 2025. Products containing these dyes may still be on shelves during compliance phase-out windows.
Is what is generally recognized as safe food actually safe for children to eat every day?
Not uniformly. Children experience a significantly higher toxicological burden from food additives than adults because of their lower body weight. According to a 2025 national study, children consume an average of 5.1 grams of food additives per day — and adjusted for body weight, their exposure is roughly twice that of adults. Specific substances like petroleum-based dyes and synthetic preservatives like BHT have been repeatedly linked to worsened ADHD symptoms, hyperactivity, and behavioral disruption in children. The MAHA Commission formally identified GRAS chemical proliferation as a driver of chronic childhood disease in May 2025.
What is the difference between a food additive and a GRAS substance when I read a label?
Both are intentionally added to food. The legal distinction is in how they were reviewed. A food additive went through formal FDA premarket approval — the agency evaluated the safety data before the ingredient could be sold. A GRAS substance bypassed that process because it was deemed safe by qualified experts, either through published science or historical use. On your ingredient label, there is no marking to tell you which is which. Both appear by their common name or chemical designation, mixed together in the same ingredient list.
Three Takeaways and One Step You Can Take Today
The GRAS system was built for common sense — to spare the FDA from reviewing table salt. What it became was a commercially optimized shortcut that allowed at least 111 unreviewed chemicals into the food supply without the government’s knowledge. The regulatory tide is shifting fast: BVO is banned, synthetic dyes are being phased out, and mandatory GRAS notification rules are imminent. But a lot of those chemicals are still in products you might buy this week.
The smartest thing you can do right now is develop the habit of checking ingredient labels — particularly for the terms “natural flavor” and “artificial flavor,” which can legally conceal unreviewed GRAS substances. Search any brand in the Huhuly search bar above to see what we have already verified.
If you want to stay current as the FDA’s GRAS reform rules roll out in 2026, subscribe to the Huhuly newsletter — we cover every regulatory update as it happens.
Reviewed by the huhuly Editorial Team
huhuly’s food transparency team reviews ingredient labels, monitors FDA regulatory updates, and tracks changes in U.S. food manufacturing. All claims are verified against official brand ingredient lists and regulatory databases before publication.
Last updated: March 2026 | Fact-checked: Yes | Sources: 20 cited
This article is for informational purposes only and does not constitute medical advice. Consult your healthcare provider before making dietary changes based on this information.
Food Dyes: What Red 40 & Yellow 5 Do to Kids
More in Food Guides
You might also find these useful




